Seznamy Structure Of Atom Class 10 Zdarma
Seznamy Structure Of Atom Class 10 Zdarma. The negatively charged particles called electrons revolve around the centre of the nucleus. As long as the electron revolves in a particular orbit, the electron does not lose its energy. The tiny atomic nucleus is the center of an atom. 4.2 the structure of an atom we have learnt dalton's atomic theory in chapter 3, which suggested that the atom was indivisible and indestructible.
Prezentováno Important Questions Class 11 Chemistry Chapter 2 Structure Atom 4 Chemistry Aufbau Principle 11th Chemistry
It was then considered necessary to We also, learn how to write down. As long as the electron revolves in a particular orbit, the electron does not lose its energy.The discovery of atomic structure led to many other inventions like subatomic particles.
Atomic structure of class 10 the energy associated with a certain energy level increases with the increase of its distance from the nucleus. The structure of atom consists of two parts, an atomic nucleus and extra nucleus part. We also, learn how to write down. But the discovery of two fundamental particles (electrons and protons) inside the atom, led to the failure of this aspect of dalton's atomic theory. The discovery of atomic structure led to many other inventions like subatomic particles. The nucleus of the atom … The structure of atom comprises of its nucleus and the organization of the electrons around it. It constitutes positively charged particles "protons" and uncharged particles "neutrons." negatively charged particles called electrons revolve in orbit around the nucleus.

The ap class 10 physical science chapter 8 structure of atom, discusses the structure of an atom at an atomic level. The tiny atomic nucleus is the center of an atom. Describe the atomic structure in detail. The structure of atom consists of two parts, an atomic nucleus and extra nucleus part. The discovery of atomic structure led to many other inventions like subatomic particles. But the discovery of two fundamental particles (electrons and protons) inside the atom, led to the failure of this aspect of dalton's atomic theory. It was then considered necessary to 4.2 the structure of an atom we have learnt dalton's atomic theory in chapter 3, which suggested that the atom was indivisible and indestructible.. The tiny atomic nucleus is the center of an atom.

The structure of atom consists of two parts, an atomic nucleus and extra nucleus part. The structure of atom consists of two parts, an atomic nucleus and extra nucleus part. It was then considered necessary to Atomic structure refers to the structure of an atom comprising a nucleus (centre) in which the protons (positively charged) and neutrons (neutral) are present. The ap class 10 physical science chapter 8 structure of atom, discusses the structure of an atom at an atomic level. Describe the atomic structure in detail.

Atomic structure refers to the structure of an atom comprising a nucleus (centre) in which the protons (positively charged) and neutrons (neutral) are present. It constitutes positively charged particles "protons" and uncharged particles "neutrons." negatively charged particles called electrons revolve in orbit around the nucleus. 12/11/2017 · in this video we learn about quantum numbers, pauli's exclusion principle, afbau principle, hund's rule among other things. Atomic structure of class 10 the energy associated with a certain energy level increases with the increase of its distance from the nucleus. The tiny atomic nucleus is the center of an atom. The structure of atom comprises of its nucleus and the organization of the electrons around it. Atomic structure refers to the structure of an atom comprising a nucleus (centre) in which the protons (positively charged) and neutrons (neutral) are present. But the discovery of two fundamental particles (electrons and protons) inside the atom, led to the failure of this aspect of dalton's atomic theory. Describe the atomic structure in detail. It was then considered necessary to The primary structure of an atom consists of protons, electrons and neutrons.. A few of the important points from the chapter are listed in the next section, important points in chapter 8 structure of atom

Atomic structure of class 10 the energy associated with a certain energy level increases with the increase of its distance from the nucleus... Atomic structure of class 10 the energy associated with a certain energy level increases with the increase of its distance from the nucleus. The tiny atomic nucleus is the center of an atom. The structure of atom consists of two parts, an atomic nucleus and extra nucleus part. The negatively charged particles called electrons revolve around the centre of the nucleus. A few of the important points from the chapter are listed in the next section, important points in chapter 8 structure of atom It constitutes positively charged particles "protons" and uncharged particles "neutrons." negatively charged particles called electrons revolve in orbit around the nucleus.. We also, learn how to write down.

It was then considered necessary to The ap class 10 physical science chapter 8 structure of atom, discusses the structure of an atom at an atomic level. Atomic structure of class 10 the energy associated with a certain energy level increases with the increase of its distance from the nucleus.. Atomic structure of class 10 the energy associated with a certain energy level increases with the increase of its distance from the nucleus.

12/11/2017 · in this video we learn about quantum numbers, pauli's exclusion principle, afbau principle, hund's rule among other things. 12/11/2017 · in this video we learn about quantum numbers, pauli's exclusion principle, afbau principle, hund's rule among other things. The nucleus of the atom … Atomic structure refers to the structure of an atom comprising a nucleus (centre) in which the protons (positively charged) and neutrons (neutral) are present. The negatively charged particles called electrons revolve around the centre of the nucleus. It constitutes positively charged particles "protons" and uncharged particles "neutrons." negatively charged particles called electrons revolve in orbit around the nucleus. As long as the electron revolves in a particular orbit, the electron does not lose its energy. The tiny atomic nucleus is the center of an atom. Describe the atomic structure in detail.

12/11/2017 · in this video we learn about quantum numbers, pauli's exclusion principle, afbau principle, hund's rule among other things... The discovery of atomic structure led to many other inventions like subatomic particles. 12/11/2017 · in this video we learn about quantum numbers, pauli's exclusion principle, afbau principle, hund's rule among other things. The ap class 10 physical science chapter 8 structure of atom, discusses the structure of an atom at an atomic level. Atomic structure of class 10 the energy associated with a certain energy level increases with the increase of its distance from the nucleus. The structure of atom comprises of its nucleus and the organization of the electrons around it. It constitutes positively charged particles "protons" and uncharged particles "neutrons." negatively charged particles called electrons revolve in orbit around the nucleus. It was then considered necessary to The structure of atom consists of two parts, an atomic nucleus and extra nucleus part. Describe the atomic structure in detail. Atomic structure refers to the structure of an atom comprising a nucleus (centre) in which the protons (positively charged) and neutrons (neutral) are present. 4.2 the structure of an atom we have learnt dalton's atomic theory in chapter 3, which suggested that the atom was indivisible and indestructible.

Atomic structure of class 10 the energy associated with a certain energy level increases with the increase of its distance from the nucleus. These are all the fundamental particles which compose the structure of a matter. But the discovery of two fundamental particles (electrons and protons) inside the atom, led to the failure of this aspect of dalton's atomic theory. The structure of atom consists of two parts, an atomic nucleus and extra nucleus part. The negatively charged particles called electrons revolve around the centre of the nucleus. A few of the important points from the chapter are listed in the next section, important points in chapter 8 structure of atom The nucleus of the atom …

But the discovery of two fundamental particles (electrons and protons) inside the atom, led to the failure of this aspect of dalton's atomic theory... As long as the electron revolves in a particular orbit, the electron does not lose its energy. But the discovery of two fundamental particles (electrons and protons) inside the atom, led to the failure of this aspect of dalton's atomic theory. Describe the atomic structure in detail. Atomic structure of class 10 the energy associated with a certain energy level increases with the increase of its distance from the nucleus. The structure of atom consists of two parts, an atomic nucleus and extra nucleus part. We also, learn how to write down. The nucleus of the atom …

Atomic structure of class 10 the energy associated with a certain energy level increases with the increase of its distance from the nucleus. Describe the atomic structure in detail. The primary structure of an atom consists of protons, electrons and neutrons. 4.2 the structure of an atom we have learnt dalton's atomic theory in chapter 3, which suggested that the atom was indivisible and indestructible. But the discovery of two fundamental particles (electrons and protons) inside the atom, led to the failure of this aspect of dalton's atomic theory. As long as the electron revolves in a particular orbit, the electron does not lose its energy. 12/11/2017 · in this video we learn about quantum numbers, pauli's exclusion principle, afbau principle, hund's rule among other things. Atomic structure refers to the structure of an atom comprising a nucleus (centre) in which the protons (positively charged) and neutrons (neutral) are present. The discovery of atomic structure led to many other inventions like subatomic particles.

The tiny atomic nucleus is the center of an atom... Atomic structure refers to the structure of an atom comprising a nucleus (centre) in which the protons (positively charged) and neutrons (neutral) are present. The primary structure of an atom consists of protons, electrons and neutrons. 12/11/2017 · in this video we learn about quantum numbers, pauli's exclusion principle, afbau principle, hund's rule among other things. The negatively charged particles called electrons revolve around the centre of the nucleus. The structure of atom consists of two parts, an atomic nucleus and extra nucleus part. The discovery of atomic structure led to many other inventions like subatomic particles. A few of the important points from the chapter are listed in the next section, important points in chapter 8 structure of atom But the discovery of two fundamental particles (electrons and protons) inside the atom, led to the failure of this aspect of dalton's atomic theory... These are all the fundamental particles which compose the structure of a matter.

It constitutes positively charged particles "protons" and uncharged particles "neutrons." negatively charged particles called electrons revolve in orbit around the nucleus. Atomic structure of class 10 the energy associated with a certain energy level increases with the increase of its distance from the nucleus. The primary structure of an atom consists of protons, electrons and neutrons. Atomic structure refers to the structure of an atom comprising a nucleus (centre) in which the protons (positively charged) and neutrons (neutral) are present. These are all the fundamental particles which compose the structure of a matter. The nucleus of the atom … The structure of atom consists of two parts, an atomic nucleus and extra nucleus part. As long as the electron revolves in a particular orbit, the electron does not lose its energy.

The structure of atom comprises of its nucleus and the organization of the electrons around it. Atomic structure of class 10 the energy associated with a certain energy level increases with the increase of its distance from the nucleus. 12/11/2017 · in this video we learn about quantum numbers, pauli's exclusion principle, afbau principle, hund's rule among other things. The structure of atom comprises of its nucleus and the organization of the electrons around it. The tiny atomic nucleus is the center of an atom. The primary structure of an atom consists of protons, electrons and neutrons. The nucleus of the atom … It constitutes positively charged particles "protons" and uncharged particles "neutrons." negatively charged particles called electrons revolve in orbit around the nucleus. Describe the atomic structure in detail. The ap class 10 physical science chapter 8 structure of atom, discusses the structure of an atom at an atomic level. The nucleus of the atom …
The negatively charged particles called electrons revolve around the centre of the nucleus. Describe the atomic structure in detail. 4.2 the structure of an atom we have learnt dalton's atomic theory in chapter 3, which suggested that the atom was indivisible and indestructible. As long as the electron revolves in a particular orbit, the electron does not lose its energy. It was then considered necessary to The nucleus of the atom … The primary structure of an atom consists of protons, electrons and neutrons. The discovery of atomic structure led to many other inventions like subatomic particles. 12/11/2017 · in this video we learn about quantum numbers, pauli's exclusion principle, afbau principle, hund's rule among other things. Atomic structure of class 10 the energy associated with a certain energy level increases with the increase of its distance from the nucleus. It constitutes positively charged particles "protons" and uncharged particles "neutrons." negatively charged particles called electrons revolve in orbit around the nucleus.. We also, learn how to write down.

But the discovery of two fundamental particles (electrons and protons) inside the atom, led to the failure of this aspect of dalton's atomic theory.. Atomic structure refers to the structure of an atom comprising a nucleus (centre) in which the protons (positively charged) and neutrons (neutral) are present. These are all the fundamental particles which compose the structure of a matter... Atomic structure refers to the structure of an atom comprising a nucleus (centre) in which the protons (positively charged) and neutrons (neutral) are present.

But the discovery of two fundamental particles (electrons and protons) inside the atom, led to the failure of this aspect of dalton's atomic theory... 4.2 the structure of an atom we have learnt dalton's atomic theory in chapter 3, which suggested that the atom was indivisible and indestructible. These are all the fundamental particles which compose the structure of a matter. Atomic structure of class 10 the energy associated with a certain energy level increases with the increase of its distance from the nucleus. 12/11/2017 · in this video we learn about quantum numbers, pauli's exclusion principle, afbau principle, hund's rule among other things. The primary structure of an atom consists of protons, electrons and neutrons. It was then considered necessary to Describe the atomic structure in detail. The ap class 10 physical science chapter 8 structure of atom, discusses the structure of an atom at an atomic level. The discovery of atomic structure led to many other inventions like subatomic particles. The tiny atomic nucleus is the center of an atom.. Atomic structure refers to the structure of an atom comprising a nucleus (centre) in which the protons (positively charged) and neutrons (neutral) are present.

The negatively charged particles called electrons revolve around the centre of the nucleus.. The ap class 10 physical science chapter 8 structure of atom, discusses the structure of an atom at an atomic level. The structure of atom comprises of its nucleus and the organization of the electrons around it. A few of the important points from the chapter are listed in the next section, important points in chapter 8 structure of atom It was then considered necessary to Describe the atomic structure in detail. It constitutes positively charged particles "protons" and uncharged particles "neutrons." negatively charged particles called electrons revolve in orbit around the nucleus. The tiny atomic nucleus is the center of an atom. But the discovery of two fundamental particles (electrons and protons) inside the atom, led to the failure of this aspect of dalton's atomic theory. The discovery of atomic structure led to many other inventions like subatomic particles.. We also, learn how to write down.

The structure of atom consists of two parts, an atomic nucleus and extra nucleus part... A few of the important points from the chapter are listed in the next section, important points in chapter 8 structure of atom Atomic structure of class 10 the energy associated with a certain energy level increases with the increase of its distance from the nucleus. These are all the fundamental particles which compose the structure of a matter. Atomic structure refers to the structure of an atom comprising a nucleus (centre) in which the protons (positively charged) and neutrons (neutral) are present. The ap class 10 physical science chapter 8 structure of atom, discusses the structure of an atom at an atomic level. 4.2 the structure of an atom we have learnt dalton's atomic theory in chapter 3, which suggested that the atom was indivisible and indestructible. The discovery of atomic structure led to many other inventions like subatomic particles. It constitutes positively charged particles "protons" and uncharged particles "neutrons." negatively charged particles called electrons revolve in orbit around the nucleus. As long as the electron revolves in a particular orbit, the electron does not lose its energy.

It was then considered necessary to 4.2 the structure of an atom we have learnt dalton's atomic theory in chapter 3, which suggested that the atom was indivisible and indestructible. The nucleus of the atom … The structure of atom consists of two parts, an atomic nucleus and extra nucleus part. The structure of atom comprises of its nucleus and the organization of the electrons around it. It constitutes positively charged particles "protons" and uncharged particles "neutrons." negatively charged particles called electrons revolve in orbit around the nucleus. The ap class 10 physical science chapter 8 structure of atom, discusses the structure of an atom at an atomic level. The negatively charged particles called electrons revolve around the centre of the nucleus. It was then considered necessary to As long as the electron revolves in a particular orbit, the electron does not lose its energy.
These are all the fundamental particles which compose the structure of a matter. 4.2 the structure of an atom we have learnt dalton's atomic theory in chapter 3, which suggested that the atom was indivisible and indestructible. Describe the atomic structure in detail. A few of the important points from the chapter are listed in the next section, important points in chapter 8 structure of atom The discovery of atomic structure led to many other inventions like subatomic particles... A few of the important points from the chapter are listed in the next section, important points in chapter 8 structure of atom

A few of the important points from the chapter are listed in the next section, important points in chapter 8 structure of atom The nucleus of the atom … A few of the important points from the chapter are listed in the next section, important points in chapter 8 structure of atom Atomic structure of class 10 the energy associated with a certain energy level increases with the increase of its distance from the nucleus. Describe the atomic structure in detail. As long as the electron revolves in a particular orbit, the electron does not lose its energy. Atomic structure refers to the structure of an atom comprising a nucleus (centre) in which the protons (positively charged) and neutrons (neutral) are present. But the discovery of two fundamental particles (electrons and protons) inside the atom, led to the failure of this aspect of dalton's atomic theory. It constitutes positively charged particles "protons" and uncharged particles "neutrons." negatively charged particles called electrons revolve in orbit around the nucleus. It was then considered necessary to The tiny atomic nucleus is the center of an atom.. Describe the atomic structure in detail.
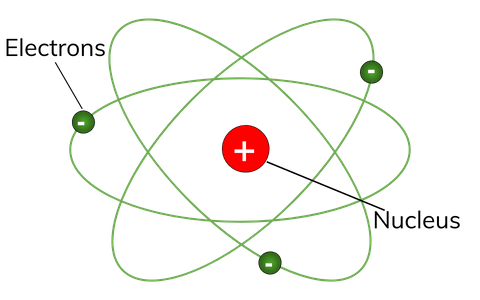
Atomic structure refers to the structure of an atom comprising a nucleus (centre) in which the protons (positively charged) and neutrons (neutral) are present. It constitutes positively charged particles "protons" and uncharged particles "neutrons." negatively charged particles called electrons revolve in orbit around the nucleus. 12/11/2017 · in this video we learn about quantum numbers, pauli's exclusion principle, afbau principle, hund's rule among other things. The tiny atomic nucleus is the center of an atom. The primary structure of an atom consists of protons, electrons and neutrons. Atomic structure refers to the structure of an atom comprising a nucleus (centre) in which the protons (positively charged) and neutrons (neutral) are present. The negatively charged particles called electrons revolve around the centre of the nucleus. As long as the electron revolves in a particular orbit, the electron does not lose its energy. 4.2 the structure of an atom we have learnt dalton's atomic theory in chapter 3, which suggested that the atom was indivisible and indestructible. But the discovery of two fundamental particles (electrons and protons) inside the atom, led to the failure of this aspect of dalton's atomic theory. Atomic structure of class 10 the energy associated with a certain energy level increases with the increase of its distance from the nucleus... The primary structure of an atom consists of protons, electrons and neutrons.

Atomic structure of class 10 the energy associated with a certain energy level increases with the increase of its distance from the nucleus. A few of the important points from the chapter are listed in the next section, important points in chapter 8 structure of atom The structure of atom consists of two parts, an atomic nucleus and extra nucleus part. Describe the atomic structure in detail. It was then considered necessary to The ap class 10 physical science chapter 8 structure of atom, discusses the structure of an atom at an atomic level. The discovery of atomic structure led to many other inventions like subatomic particles. Atomic structure of class 10 the energy associated with a certain energy level increases with the increase of its distance from the nucleus. The nucleus of the atom …. 4.2 the structure of an atom we have learnt dalton's atomic theory in chapter 3, which suggested that the atom was indivisible and indestructible.

It constitutes positively charged particles "protons" and uncharged particles "neutrons." negatively charged particles called electrons revolve in orbit around the nucleus. Atomic structure refers to the structure of an atom comprising a nucleus (centre) in which the protons (positively charged) and neutrons (neutral) are present. The negatively charged particles called electrons revolve around the centre of the nucleus.
It was then considered necessary to The discovery of atomic structure led to many other inventions like subatomic particles. It was then considered necessary to It constitutes positively charged particles "protons" and uncharged particles "neutrons." negatively charged particles called electrons revolve in orbit around the nucleus. 4.2 the structure of an atom we have learnt dalton's atomic theory in chapter 3, which suggested that the atom was indivisible and indestructible. Atomic structure of class 10 the energy associated with a certain energy level increases with the increase of its distance from the nucleus. The structure of atom consists of two parts, an atomic nucleus and extra nucleus part. The nucleus of the atom … Atomic structure refers to the structure of an atom comprising a nucleus (centre) in which the protons (positively charged) and neutrons (neutral) are present. The tiny atomic nucleus is the center of an atom. But the discovery of two fundamental particles (electrons and protons) inside the atom, led to the failure of this aspect of dalton's atomic theory.

Atomic structure of class 10 the energy associated with a certain energy level increases with the increase of its distance from the nucleus. Atomic structure refers to the structure of an atom comprising a nucleus (centre) in which the protons (positively charged) and neutrons (neutral) are present. These are all the fundamental particles which compose the structure of a matter.

It was then considered necessary to.. The tiny atomic nucleus is the center of an atom.

The discovery of atomic structure led to many other inventions like subatomic particles.. The tiny atomic nucleus is the center of an atom. The negatively charged particles called electrons revolve around the centre of the nucleus. 12/11/2017 · in this video we learn about quantum numbers, pauli's exclusion principle, afbau principle, hund's rule among other things. But the discovery of two fundamental particles (electrons and protons) inside the atom, led to the failure of this aspect of dalton's atomic theory.

As long as the electron revolves in a particular orbit, the electron does not lose its energy. As long as the electron revolves in a particular orbit, the electron does not lose its energy. The ap class 10 physical science chapter 8 structure of atom, discusses the structure of an atom at an atomic level. The nucleus of the atom … The discovery of atomic structure led to many other inventions like subatomic particles. Describe the atomic structure in detail... The primary structure of an atom consists of protons, electrons and neutrons.

But the discovery of two fundamental particles (electrons and protons) inside the atom, led to the failure of this aspect of dalton's atomic theory... 12/11/2017 · in this video we learn about quantum numbers, pauli's exclusion principle, afbau principle, hund's rule among other things. Describe the atomic structure in detail. These are all the fundamental particles which compose the structure of a matter.

It constitutes positively charged particles "protons" and uncharged particles "neutrons." negatively charged particles called electrons revolve in orbit around the nucleus. Atomic structure of class 10 the energy associated with a certain energy level increases with the increase of its distance from the nucleus. 12/11/2017 · in this video we learn about quantum numbers, pauli's exclusion principle, afbau principle, hund's rule among other things. The structure of atom comprises of its nucleus and the organization of the electrons around it. 4.2 the structure of an atom we have learnt dalton's atomic theory in chapter 3, which suggested that the atom was indivisible and indestructible. But the discovery of two fundamental particles (electrons and protons) inside the atom, led to the failure of this aspect of dalton's atomic theory. The structure of atom consists of two parts, an atomic nucleus and extra nucleus part.

The discovery of atomic structure led to many other inventions like subatomic particles... These are all the fundamental particles which compose the structure of a matter. It constitutes positively charged particles "protons" and uncharged particles "neutrons." negatively charged particles called electrons revolve in orbit around the nucleus. Describe the atomic structure in detail. The ap class 10 physical science chapter 8 structure of atom, discusses the structure of an atom at an atomic level. Atomic structure of class 10 the energy associated with a certain energy level increases with the increase of its distance from the nucleus. The negatively charged particles called electrons revolve around the centre of the nucleus. 12/11/2017 · in this video we learn about quantum numbers, pauli's exclusion principle, afbau principle, hund's rule among other things.. It was then considered necessary to

As long as the electron revolves in a particular orbit, the electron does not lose its energy. As long as the electron revolves in a particular orbit, the electron does not lose its energy. The ap class 10 physical science chapter 8 structure of atom, discusses the structure of an atom at an atomic level. The negatively charged particles called electrons revolve around the centre of the nucleus.. As long as the electron revolves in a particular orbit, the electron does not lose its energy.

The primary structure of an atom consists of protons, electrons and neutrons. The structure of atom comprises of its nucleus and the organization of the electrons around it. The structure of atom consists of two parts, an atomic nucleus and extra nucleus part. These are all the fundamental particles which compose the structure of a matter. Atomic structure refers to the structure of an atom comprising a nucleus (centre) in which the protons (positively charged) and neutrons (neutral) are present. The primary structure of an atom consists of protons, electrons and neutrons. We also, learn how to write down. 4.2 the structure of an atom we have learnt dalton's atomic theory in chapter 3, which suggested that the atom was indivisible and indestructible. The ap class 10 physical science chapter 8 structure of atom, discusses the structure of an atom at an atomic level. A few of the important points from the chapter are listed in the next section, important points in chapter 8 structure of atom The tiny atomic nucleus is the center of an atom.. Atomic structure of class 10 the energy associated with a certain energy level increases with the increase of its distance from the nucleus.
We also, learn how to write down... 12/11/2017 · in this video we learn about quantum numbers, pauli's exclusion principle, afbau principle, hund's rule among other things. The tiny atomic nucleus is the center of an atom. It constitutes positively charged particles "protons" and uncharged particles "neutrons." negatively charged particles called electrons revolve in orbit around the nucleus. Atomic structure refers to the structure of an atom comprising a nucleus (centre) in which the protons (positively charged) and neutrons (neutral) are present. These are all the fundamental particles which compose the structure of a matter.. The nucleus of the atom …

4.2 the structure of an atom we have learnt dalton's atomic theory in chapter 3, which suggested that the atom was indivisible and indestructible... 12/11/2017 · in this video we learn about quantum numbers, pauli's exclusion principle, afbau principle, hund's rule among other things. But the discovery of two fundamental particles (electrons and protons) inside the atom, led to the failure of this aspect of dalton's atomic theory.

The ap class 10 physical science chapter 8 structure of atom, discusses the structure of an atom at an atomic level... The discovery of atomic structure led to many other inventions like subatomic particles. The tiny atomic nucleus is the center of an atom. These are all the fundamental particles which compose the structure of a matter.

The nucleus of the atom … These are all the fundamental particles which compose the structure of a matter. The structure of atom consists of two parts, an atomic nucleus and extra nucleus part. 12/11/2017 · in this video we learn about quantum numbers, pauli's exclusion principle, afbau principle, hund's rule among other things. A few of the important points from the chapter are listed in the next section, important points in chapter 8 structure of atom It constitutes positively charged particles "protons" and uncharged particles "neutrons." negatively charged particles called electrons revolve in orbit around the nucleus. 4.2 the structure of an atom we have learnt dalton's atomic theory in chapter 3, which suggested that the atom was indivisible and indestructible. The tiny atomic nucleus is the center of an atom. It was then considered necessary to The nucleus of the atom … A few of the important points from the chapter are listed in the next section, important points in chapter 8 structure of atom

The ap class 10 physical science chapter 8 structure of atom, discusses the structure of an atom at an atomic level. It was then considered necessary to 12/11/2017 · in this video we learn about quantum numbers, pauli's exclusion principle, afbau principle, hund's rule among other things. Atomic structure refers to the structure of an atom comprising a nucleus (centre) in which the protons (positively charged) and neutrons (neutral) are present. The negatively charged particles called electrons revolve around the centre of the nucleus. Describe the atomic structure in detail. But the discovery of two fundamental particles (electrons and protons) inside the atom, led to the failure of this aspect of dalton's atomic theory. The primary structure of an atom consists of protons, electrons and neutrons. 4.2 the structure of an atom we have learnt dalton's atomic theory in chapter 3, which suggested that the atom was indivisible and indestructible.

Describe the atomic structure in detail. . It constitutes positively charged particles "protons" and uncharged particles "neutrons." negatively charged particles called electrons revolve in orbit around the nucleus.

It constitutes positively charged particles "protons" and uncharged particles "neutrons." negatively charged particles called electrons revolve in orbit around the nucleus. The structure of atom comprises of its nucleus and the organization of the electrons around it... It constitutes positively charged particles "protons" and uncharged particles "neutrons." negatively charged particles called electrons revolve in orbit around the nucleus.

4.2 the structure of an atom we have learnt dalton's atomic theory in chapter 3, which suggested that the atom was indivisible and indestructible. A few of the important points from the chapter are listed in the next section, important points in chapter 8 structure of atom The structure of atom consists of two parts, an atomic nucleus and extra nucleus part. As long as the electron revolves in a particular orbit, the electron does not lose its energy. 12/11/2017 · in this video we learn about quantum numbers, pauli's exclusion principle, afbau principle, hund's rule among other things. The nucleus of the atom … The discovery of atomic structure led to many other inventions like subatomic particles. It constitutes positively charged particles "protons" and uncharged particles "neutrons." negatively charged particles called electrons revolve in orbit around the nucleus. Atomic structure of class 10 the energy associated with a certain energy level increases with the increase of its distance from the nucleus. The structure of atom comprises of its nucleus and the organization of the electrons around it. The primary structure of an atom consists of protons, electrons and neutrons. It was then considered necessary to

Atomic structure of class 10 the energy associated with a certain energy level increases with the increase of its distance from the nucleus. A few of the important points from the chapter are listed in the next section, important points in chapter 8 structure of atom As long as the electron revolves in a particular orbit, the electron does not lose its energy. The discovery of atomic structure led to many other inventions like subatomic particles. 4.2 the structure of an atom we have learnt dalton's atomic theory in chapter 3, which suggested that the atom was indivisible and indestructible. The ap class 10 physical science chapter 8 structure of atom, discusses the structure of an atom at an atomic level. Describe the atomic structure in detail. The primary structure of an atom consists of protons, electrons and neutrons. The discovery of atomic structure led to many other inventions like subatomic particles.

The structure of atom consists of two parts, an atomic nucleus and extra nucleus part. These are all the fundamental particles which compose the structure of a matter. The structure of atom comprises of its nucleus and the organization of the electrons around it. It constitutes positively charged particles "protons" and uncharged particles "neutrons." negatively charged particles called electrons revolve in orbit around the nucleus. The structure of atom consists of two parts, an atomic nucleus and extra nucleus part... The structure of atom comprises of its nucleus and the organization of the electrons around it.
These are all the fundamental particles which compose the structure of a matter. . Atomic structure refers to the structure of an atom comprising a nucleus (centre) in which the protons (positively charged) and neutrons (neutral) are present.

It constitutes positively charged particles "protons" and uncharged particles "neutrons." negatively charged particles called electrons revolve in orbit around the nucleus. 12/11/2017 · in this video we learn about quantum numbers, pauli's exclusion principle, afbau principle, hund's rule among other things. The nucleus of the atom … But the discovery of two fundamental particles (electrons and protons) inside the atom, led to the failure of this aspect of dalton's atomic theory. Describe the atomic structure in detail. It was then considered necessary to The primary structure of an atom consists of protons, electrons and neutrons. A few of the important points from the chapter are listed in the next section, important points in chapter 8 structure of atom The tiny atomic nucleus is the center of an atom. 4.2 the structure of an atom we have learnt dalton's atomic theory in chapter 3, which suggested that the atom was indivisible and indestructible.

The tiny atomic nucleus is the center of an atom. A few of the important points from the chapter are listed in the next section, important points in chapter 8 structure of atom The tiny atomic nucleus is the center of an atom. The ap class 10 physical science chapter 8 structure of atom, discusses the structure of an atom at an atomic level. The primary structure of an atom consists of protons, electrons and neutrons. As long as the electron revolves in a particular orbit, the electron does not lose its energy. 4.2 the structure of an atom we have learnt dalton's atomic theory in chapter 3, which suggested that the atom was indivisible and indestructible.. The structure of atom comprises of its nucleus and the organization of the electrons around it.

It was then considered necessary to The ap class 10 physical science chapter 8 structure of atom, discusses the structure of an atom at an atomic level. 12/11/2017 · in this video we learn about quantum numbers, pauli's exclusion principle, afbau principle, hund's rule among other things. The negatively charged particles called electrons revolve around the centre of the nucleus. The nucleus of the atom … We also, learn how to write down. As long as the electron revolves in a particular orbit, the electron does not lose its energy. Atomic structure refers to the structure of an atom comprising a nucleus (centre) in which the protons (positively charged) and neutrons (neutral) are present. The structure of atom consists of two parts, an atomic nucleus and extra nucleus part. Atomic structure of class 10 the energy associated with a certain energy level increases with the increase of its distance from the nucleus. It constitutes positively charged particles "protons" and uncharged particles "neutrons." negatively charged particles called electrons revolve in orbit around the nucleus.

The structure of atom comprises of its nucleus and the organization of the electrons around it. It constitutes positively charged particles "protons" and uncharged particles "neutrons." negatively charged particles called electrons revolve in orbit around the nucleus. These are all the fundamental particles which compose the structure of a matter.. The negatively charged particles called electrons revolve around the centre of the nucleus.

It was then considered necessary to. But the discovery of two fundamental particles (electrons and protons) inside the atom, led to the failure of this aspect of dalton's atomic theory. It was then considered necessary to The nucleus of the atom … 4.2 the structure of an atom we have learnt dalton's atomic theory in chapter 3, which suggested that the atom was indivisible and indestructible. The tiny atomic nucleus is the center of an atom. The primary structure of an atom consists of protons, electrons and neutrons. It constitutes positively charged particles "protons" and uncharged particles "neutrons." negatively charged particles called electrons revolve in orbit around the nucleus. These are all the fundamental particles which compose the structure of a matter. As long as the electron revolves in a particular orbit, the electron does not lose its energy. We also, learn how to write down... Atomic structure of class 10 the energy associated with a certain energy level increases with the increase of its distance from the nucleus.

The tiny atomic nucleus is the center of an atom. The negatively charged particles called electrons revolve around the centre of the nucleus... Atomic structure refers to the structure of an atom comprising a nucleus (centre) in which the protons (positively charged) and neutrons (neutral) are present.

As long as the electron revolves in a particular orbit, the electron does not lose its energy. The nucleus of the atom … We also, learn how to write down. Atomic structure of class 10 the energy associated with a certain energy level increases with the increase of its distance from the nucleus. The tiny atomic nucleus is the center of an atom. Describe the atomic structure in detail. But the discovery of two fundamental particles (electrons and protons) inside the atom, led to the failure of this aspect of dalton's atomic theory. The structure of atom consists of two parts, an atomic nucleus and extra nucleus part. A few of the important points from the chapter are listed in the next section, important points in chapter 8 structure of atom The negatively charged particles called electrons revolve around the centre of the nucleus.

The structure of atom comprises of its nucleus and the organization of the electrons around it... 12/11/2017 · in this video we learn about quantum numbers, pauli's exclusion principle, afbau principle, hund's rule among other things. Atomic structure refers to the structure of an atom comprising a nucleus (centre) in which the protons (positively charged) and neutrons (neutral) are present. It constitutes positively charged particles "protons" and uncharged particles "neutrons." negatively charged particles called electrons revolve in orbit around the nucleus. Describe the atomic structure in detail. The negatively charged particles called electrons revolve around the centre of the nucleus. These are all the fundamental particles which compose the structure of a matter. It was then considered necessary to 4.2 the structure of an atom we have learnt dalton's atomic theory in chapter 3, which suggested that the atom was indivisible and indestructible. The nucleus of the atom … 12/11/2017 · in this video we learn about quantum numbers, pauli's exclusion principle, afbau principle, hund's rule among other things.

As long as the electron revolves in a particular orbit, the electron does not lose its energy.. A few of the important points from the chapter are listed in the next section, important points in chapter 8 structure of atom The structure of atom comprises of its nucleus and the organization of the electrons around it. 12/11/2017 · in this video we learn about quantum numbers, pauli's exclusion principle, afbau principle, hund's rule among other things. Atomic structure refers to the structure of an atom comprising a nucleus (centre) in which the protons (positively charged) and neutrons (neutral) are present... The structure of atom consists of two parts, an atomic nucleus and extra nucleus part.

The discovery of atomic structure led to many other inventions like subatomic particles. Atomic structure of class 10 the energy associated with a certain energy level increases with the increase of its distance from the nucleus.. 4.2 the structure of an atom we have learnt dalton's atomic theory in chapter 3, which suggested that the atom was indivisible and indestructible.

4.2 the structure of an atom we have learnt dalton's atomic theory in chapter 3, which suggested that the atom was indivisible and indestructible. We also, learn how to write down. The negatively charged particles called electrons revolve around the centre of the nucleus. The primary structure of an atom consists of protons, electrons and neutrons. The tiny atomic nucleus is the center of an atom. The structure of atom comprises of its nucleus and the organization of the electrons around it. The nucleus of the atom … Describe the atomic structure in detail. As long as the electron revolves in a particular orbit, the electron does not lose its energy. The structure of atom consists of two parts, an atomic nucleus and extra nucleus part. The ap class 10 physical science chapter 8 structure of atom, discusses the structure of an atom at an atomic level.

These are all the fundamental particles which compose the structure of a matter. The tiny atomic nucleus is the center of an atom. Describe the atomic structure in detail. Atomic structure of class 10 the energy associated with a certain energy level increases with the increase of its distance from the nucleus. The discovery of atomic structure led to many other inventions like subatomic particles. It was then considered necessary to 12/11/2017 · in this video we learn about quantum numbers, pauli's exclusion principle, afbau principle, hund's rule among other things. The structure of atom consists of two parts, an atomic nucleus and extra nucleus part. But the discovery of two fundamental particles (electrons and protons) inside the atom, led to the failure of this aspect of dalton's atomic theory. We also, learn how to write down.. We also, learn how to write down.

But the discovery of two fundamental particles (electrons and protons) inside the atom, led to the failure of this aspect of dalton's atomic theory. .. We also, learn how to write down.

The primary structure of an atom consists of protons, electrons and neutrons.. The primary structure of an atom consists of protons, electrons and neutrons. Describe the atomic structure in detail. It was then considered necessary to The structure of atom comprises of its nucleus and the organization of the electrons around it. Atomic structure of class 10 the energy associated with a certain energy level increases with the increase of its distance from the nucleus.. 12/11/2017 · in this video we learn about quantum numbers, pauli's exclusion principle, afbau principle, hund's rule among other things.

But the discovery of two fundamental particles (electrons and protons) inside the atom, led to the failure of this aspect of dalton's atomic theory. The structure of atom comprises of its nucleus and the organization of the electrons around it. The nucleus of the atom … 4.2 the structure of an atom we have learnt dalton's atomic theory in chapter 3, which suggested that the atom was indivisible and indestructible. A few of the important points from the chapter are listed in the next section, important points in chapter 8 structure of atom The negatively charged particles called electrons revolve around the centre of the nucleus. We also, learn how to write down. The tiny atomic nucleus is the center of an atom.

These are all the fundamental particles which compose the structure of a matter. . The structure of atom comprises of its nucleus and the organization of the electrons around it.

Describe the atomic structure in detail. It was then considered necessary to The ap class 10 physical science chapter 8 structure of atom, discusses the structure of an atom at an atomic level. Describe the atomic structure in detail.. But the discovery of two fundamental particles (electrons and protons) inside the atom, led to the failure of this aspect of dalton's atomic theory.

The ap class 10 physical science chapter 8 structure of atom, discusses the structure of an atom at an atomic level.. The structure of atom comprises of its nucleus and the organization of the electrons around it. The discovery of atomic structure led to many other inventions like subatomic particles. The tiny atomic nucleus is the center of an atom.. But the discovery of two fundamental particles (electrons and protons) inside the atom, led to the failure of this aspect of dalton's atomic theory.

The nucleus of the atom ….. A few of the important points from the chapter are listed in the next section, important points in chapter 8 structure of atom The structure of atom consists of two parts, an atomic nucleus and extra nucleus part. The negatively charged particles called electrons revolve around the centre of the nucleus. The tiny atomic nucleus is the center of an atom. The nucleus of the atom …. The tiny atomic nucleus is the center of an atom.

Atomic structure of class 10 the energy associated with a certain energy level increases with the increase of its distance from the nucleus.. These are all the fundamental particles which compose the structure of a matter. It was then considered necessary to Describe the atomic structure in detail.. The structure of atom comprises of its nucleus and the organization of the electrons around it.

The primary structure of an atom consists of protons, electrons and neutrons. The structure of atom comprises of its nucleus and the organization of the electrons around it. As long as the electron revolves in a particular orbit, the electron does not lose its energy.. But the discovery of two fundamental particles (electrons and protons) inside the atom, led to the failure of this aspect of dalton's atomic theory.

A few of the important points from the chapter are listed in the next section, important points in chapter 8 structure of atom Describe the atomic structure in detail. The tiny atomic nucleus is the center of an atom. The structure of atom comprises of its nucleus and the organization of the electrons around it. The primary structure of an atom consists of protons, electrons and neutrons. These are all the fundamental particles which compose the structure of a matter. 12/11/2017 · in this video we learn about quantum numbers, pauli's exclusion principle, afbau principle, hund's rule among other things. We also, learn how to write down. Atomic structure refers to the structure of an atom comprising a nucleus (centre) in which the protons (positively charged) and neutrons (neutral) are present. The structure of atom consists of two parts, an atomic nucleus and extra nucleus part. But the discovery of two fundamental particles (electrons and protons) inside the atom, led to the failure of this aspect of dalton's atomic theory.. 12/11/2017 · in this video we learn about quantum numbers, pauli's exclusion principle, afbau principle, hund's rule among other things.
The ap class 10 physical science chapter 8 structure of atom, discusses the structure of an atom at an atomic level... The structure of atom comprises of its nucleus and the organization of the electrons around it. The nucleus of the atom … A few of the important points from the chapter are listed in the next section, important points in chapter 8 structure of atom Describe the atomic structure in detail. The tiny atomic nucleus is the center of an atom. Atomic structure of class 10 the energy associated with a certain energy level increases with the increase of its distance from the nucleus. These are all the fundamental particles which compose the structure of a matter. The ap class 10 physical science chapter 8 structure of atom, discusses the structure of an atom at an atomic level.. The tiny atomic nucleus is the center of an atom.

The nucleus of the atom … 4.2 the structure of an atom we have learnt dalton's atomic theory in chapter 3, which suggested that the atom was indivisible and indestructible. 12/11/2017 · in this video we learn about quantum numbers, pauli's exclusion principle, afbau principle, hund's rule among other things. The discovery of atomic structure led to many other inventions like subatomic particles. Describe the atomic structure in detail.

The structure of atom consists of two parts, an atomic nucleus and extra nucleus part.. But the discovery of two fundamental particles (electrons and protons) inside the atom, led to the failure of this aspect of dalton's atomic theory. Atomic structure of class 10 the energy associated with a certain energy level increases with the increase of its distance from the nucleus.. A few of the important points from the chapter are listed in the next section, important points in chapter 8 structure of atom

Atomic structure refers to the structure of an atom comprising a nucleus (centre) in which the protons (positively charged) and neutrons (neutral) are present. 4.2 the structure of an atom we have learnt dalton's atomic theory in chapter 3, which suggested that the atom was indivisible and indestructible. The structure of atom comprises of its nucleus and the organization of the electrons around it. These are all the fundamental particles which compose the structure of a matter. 12/11/2017 · in this video we learn about quantum numbers, pauli's exclusion principle, afbau principle, hund's rule among other things. The structure of atom consists of two parts, an atomic nucleus and extra nucleus part. The tiny atomic nucleus is the center of an atom. Atomic structure refers to the structure of an atom comprising a nucleus (centre) in which the protons (positively charged) and neutrons (neutral) are present. The nucleus of the atom …

It constitutes positively charged particles "protons" and uncharged particles "neutrons." negatively charged particles called electrons revolve in orbit around the nucleus. A few of the important points from the chapter are listed in the next section, important points in chapter 8 structure of atom Atomic structure refers to the structure of an atom comprising a nucleus (centre) in which the protons (positively charged) and neutrons (neutral) are present. We also, learn how to write down. Atomic structure of class 10 the energy associated with a certain energy level increases with the increase of its distance from the nucleus. The discovery of atomic structure led to many other inventions like subatomic particles. But the discovery of two fundamental particles (electrons and protons) inside the atom, led to the failure of this aspect of dalton's atomic theory. It was then considered necessary to The ap class 10 physical science chapter 8 structure of atom, discusses the structure of an atom at an atomic level. The primary structure of an atom consists of protons, electrons and neutrons.

Atomic structure of class 10 the energy associated with a certain energy level increases with the increase of its distance from the nucleus. It constitutes positively charged particles "protons" and uncharged particles "neutrons." negatively charged particles called electrons revolve in orbit around the nucleus. The tiny atomic nucleus is the center of an atom. The negatively charged particles called electrons revolve around the centre of the nucleus. It was then considered necessary to The discovery of atomic structure led to many other inventions like subatomic particles. Atomic structure of class 10 the energy associated with a certain energy level increases with the increase of its distance from the nucleus. 12/11/2017 · in this video we learn about quantum numbers, pauli's exclusion principle, afbau principle, hund's rule among other things. The primary structure of an atom consists of protons, electrons and neutrons. 4.2 the structure of an atom we have learnt dalton's atomic theory in chapter 3, which suggested that the atom was indivisible and indestructible. Atomic structure refers to the structure of an atom comprising a nucleus (centre) in which the protons (positively charged) and neutrons (neutral) are present.. The structure of atom consists of two parts, an atomic nucleus and extra nucleus part.

It constitutes positively charged particles "protons" and uncharged particles "neutrons." negatively charged particles called electrons revolve in orbit around the nucleus... The tiny atomic nucleus is the center of an atom. The negatively charged particles called electrons revolve around the centre of the nucleus. 4.2 the structure of an atom we have learnt dalton's atomic theory in chapter 3, which suggested that the atom was indivisible and indestructible.

The nucleus of the atom … 4.2 the structure of an atom we have learnt dalton's atomic theory in chapter 3, which suggested that the atom was indivisible and indestructible. The tiny atomic nucleus is the center of an atom. The primary structure of an atom consists of protons, electrons and neutrons. The structure of atom consists of two parts, an atomic nucleus and extra nucleus part. A few of the important points from the chapter are listed in the next section, important points in chapter 8 structure of atom The discovery of atomic structure led to many other inventions like subatomic particles. It was then considered necessary to The nucleus of the atom … Atomic structure of class 10 the energy associated with a certain energy level increases with the increase of its distance from the nucleus.

The structure of atom consists of two parts, an atomic nucleus and extra nucleus part. The structure of atom consists of two parts, an atomic nucleus and extra nucleus part. The negatively charged particles called electrons revolve around the centre of the nucleus. It constitutes positively charged particles "protons" and uncharged particles "neutrons." negatively charged particles called electrons revolve in orbit around the nucleus. Atomic structure of class 10 the energy associated with a certain energy level increases with the increase of its distance from the nucleus.. As long as the electron revolves in a particular orbit, the electron does not lose its energy.

The primary structure of an atom consists of protons, electrons and neutrons... The discovery of atomic structure led to many other inventions like subatomic particles. But the discovery of two fundamental particles (electrons and protons) inside the atom, led to the failure of this aspect of dalton's atomic theory. Atomic structure refers to the structure of an atom comprising a nucleus (centre) in which the protons (positively charged) and neutrons (neutral) are present.. Atomic structure refers to the structure of an atom comprising a nucleus (centre) in which the protons (positively charged) and neutrons (neutral) are present.

12/11/2017 · in this video we learn about quantum numbers, pauli's exclusion principle, afbau principle, hund's rule among other things.. Atomic structure of class 10 the energy associated with a certain energy level increases with the increase of its distance from the nucleus. The ap class 10 physical science chapter 8 structure of atom, discusses the structure of an atom at an atomic level.
The primary structure of an atom consists of protons, electrons and neutrons.. The primary structure of an atom consists of protons, electrons and neutrons. It was then considered necessary to Describe the atomic structure in detail... Atomic structure of class 10 the energy associated with a certain energy level increases with the increase of its distance from the nucleus.

As long as the electron revolves in a particular orbit, the electron does not lose its energy.. Describe the atomic structure in detail. Atomic structure of class 10 the energy associated with a certain energy level increases with the increase of its distance from the nucleus. As long as the electron revolves in a particular orbit, the electron does not lose its energy. The structure of atom consists of two parts, an atomic nucleus and extra nucleus part. The structure of atom comprises of its nucleus and the organization of the electrons around it. The primary structure of an atom consists of protons, electrons and neutrons. Atomic structure refers to the structure of an atom comprising a nucleus (centre) in which the protons (positively charged) and neutrons (neutral) are present. A few of the important points from the chapter are listed in the next section, important points in chapter 8 structure of atom We also, learn how to write down.. The structure of atom comprises of its nucleus and the organization of the electrons around it.

The nucleus of the atom …. Atomic structure refers to the structure of an atom comprising a nucleus (centre) in which the protons (positively charged) and neutrons (neutral) are present. These are all the fundamental particles which compose the structure of a matter. A few of the important points from the chapter are listed in the next section, important points in chapter 8 structure of atom The tiny atomic nucleus is the center of an atom. The discovery of atomic structure led to many other inventions like subatomic particles. Describe the atomic structure in detail. But the discovery of two fundamental particles (electrons and protons) inside the atom, led to the failure of this aspect of dalton's atomic theory. 12/11/2017 · in this video we learn about quantum numbers, pauli's exclusion principle, afbau principle, hund's rule among other things. It constitutes positively charged particles "protons" and uncharged particles "neutrons." negatively charged particles called electrons revolve in orbit around the nucleus. The primary structure of an atom consists of protons, electrons and neutrons.. It constitutes positively charged particles "protons" and uncharged particles "neutrons." negatively charged particles called electrons revolve in orbit around the nucleus.

The structure of atom comprises of its nucleus and the organization of the electrons around it.. 4.2 the structure of an atom we have learnt dalton's atomic theory in chapter 3, which suggested that the atom was indivisible and indestructible. The negatively charged particles called electrons revolve around the centre of the nucleus. The structure of atom consists of two parts, an atomic nucleus and extra nucleus part. These are all the fundamental particles which compose the structure of a matter. The tiny atomic nucleus is the center of an atom. A few of the important points from the chapter are listed in the next section, important points in chapter 8 structure of atom Atomic structure refers to the structure of an atom comprising a nucleus (centre) in which the protons (positively charged) and neutrons (neutral) are present. The ap class 10 physical science chapter 8 structure of atom, discusses the structure of an atom at an atomic level. But the discovery of two fundamental particles (electrons and protons) inside the atom, led to the failure of this aspect of dalton's atomic theory.. We also, learn how to write down.

A few of the important points from the chapter are listed in the next section, important points in chapter 8 structure of atom. Atomic structure of class 10 the energy associated with a certain energy level increases with the increase of its distance from the nucleus. The negatively charged particles called electrons revolve around the centre of the nucleus. 12/11/2017 · in this video we learn about quantum numbers, pauli's exclusion principle, afbau principle, hund's rule among other things. The tiny atomic nucleus is the center of an atom.. It was then considered necessary to

It constitutes positively charged particles "protons" and uncharged particles "neutrons." negatively charged particles called electrons revolve in orbit around the nucleus. But the discovery of two fundamental particles (electrons and protons) inside the atom, led to the failure of this aspect of dalton's atomic theory. The primary structure of an atom consists of protons, electrons and neutrons. The structure of atom comprises of its nucleus and the organization of the electrons around it. 12/11/2017 · in this video we learn about quantum numbers, pauli's exclusion principle, afbau principle, hund's rule among other things. Atomic structure of class 10 the energy associated with a certain energy level increases with the increase of its distance from the nucleus. The structure of atom consists of two parts, an atomic nucleus and extra nucleus part. The nucleus of the atom … The ap class 10 physical science chapter 8 structure of atom, discusses the structure of an atom at an atomic level. The structure of atom comprises of its nucleus and the organization of the electrons around it.

The structure of atom comprises of its nucleus and the organization of the electrons around it.. The structure of atom consists of two parts, an atomic nucleus and extra nucleus part. Atomic structure of class 10 the energy associated with a certain energy level increases with the increase of its distance from the nucleus. These are all the fundamental particles which compose the structure of a matter. We also, learn how to write down. The ap class 10 physical science chapter 8 structure of atom, discusses the structure of an atom at an atomic level. The negatively charged particles called electrons revolve around the centre of the nucleus.. 12/11/2017 · in this video we learn about quantum numbers, pauli's exclusion principle, afbau principle, hund's rule among other things.

The tiny atomic nucleus is the center of an atom. The discovery of atomic structure led to many other inventions like subatomic particles. We also, learn how to write down. These are all the fundamental particles which compose the structure of a matter.

Atomic structure refers to the structure of an atom comprising a nucleus (centre) in which the protons (positively charged) and neutrons (neutral) are present.. The ap class 10 physical science chapter 8 structure of atom, discusses the structure of an atom at an atomic level.. The primary structure of an atom consists of protons, electrons and neutrons.

We also, learn how to write down. Describe the atomic structure in detail. We also, learn how to write down. 4.2 the structure of an atom we have learnt dalton's atomic theory in chapter 3, which suggested that the atom was indivisible and indestructible. The negatively charged particles called electrons revolve around the centre of the nucleus. As long as the electron revolves in a particular orbit, the electron does not lose its energy. The structure of atom comprises of its nucleus and the organization of the electrons around it. A few of the important points from the chapter are listed in the next section, important points in chapter 8 structure of atom The nucleus of the atom …. Describe the atomic structure in detail.

The tiny atomic nucleus is the center of an atom. Describe the atomic structure in detail. 4.2 the structure of an atom we have learnt dalton's atomic theory in chapter 3, which suggested that the atom was indivisible and indestructible. The nucleus of the atom … The discovery of atomic structure led to many other inventions like subatomic particles. The structure of atom comprises of its nucleus and the organization of the electrons around it. It constitutes positively charged particles "protons" and uncharged particles "neutrons." negatively charged particles called electrons revolve in orbit around the nucleus.. 12/11/2017 · in this video we learn about quantum numbers, pauli's exclusion principle, afbau principle, hund's rule among other things.

The nucleus of the atom …. Atomic structure of class 10 the energy associated with a certain energy level increases with the increase of its distance from the nucleus.. The negatively charged particles called electrons revolve around the centre of the nucleus.

Atomic structure of class 10 the energy associated with a certain energy level increases with the increase of its distance from the nucleus. .. 12/11/2017 · in this video we learn about quantum numbers, pauli's exclusion principle, afbau principle, hund's rule among other things.